The Health Secretary has formally asked the medicines regulator to assess the Pfizer/BioNTech coronavirus vaccine for use in the UK.
Matt Hancock hailed it as “another important step forward in tackling this pandemic”.
But he said while the regulator’s approval of a vaccine would see a rollout ready to start next month, there is “still a long way to go”.
– Who is developing the coronavirus vaccines?
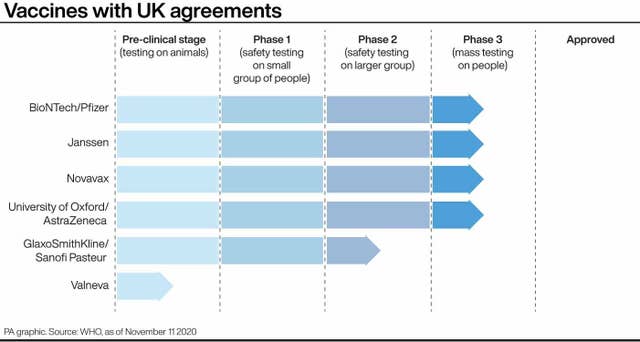
The front-runners in the Covid-19 vaccine race are US pharmaceutical firm Pfizer, which is working with German biotech company BioNTech, Oxford University, working with AstraZeneca, and US company Moderna.
Interim trial results for Moderna’s jab have suggested it may be 94.5% effective against Covid-19.
Pfizer/BioNTech has released data suggesting their vaccine is 95% effective in preventing Covid-19.

Further results showing its effectiveness are expected to be released in the coming weeks.
– When will the vaccines be ready?
Mr Hancock said if the Medicines and Healthcare products Regulatory Agency (MHRA) approves a vaccine the rollout could begin in December.
But he cautioned that the speed of the roll-out would depend on the speed it could be manufactured.
“We are heading in the right direction but there is still a long way to go.”
Sir John Bell, who is part of the Oxford vaccine team, said he expects two or three jabs to be available by the new year.
– What is the MHRA?
The MHRA is an executive agency of the Department of Health and Social Care.
Among its responsibilities are making sure that medicines meet applicable standards of safety, quality and efficacy, and ensuring the supply chain for those medicines is safe and secure.
The agency employs more than 1,200 people, with facilities across London, York and Hertfordshire.
– How long will the vaccine approval process take?
Mr Hancock has stressed the independence of the MHRA and said he did not want to “pre-judge”.
Asked what assurances he had been given by the medicines regulator over how long its process takes, he said: “It is absolutely a question for them how long they take, I’m not going to pre-judge that at all.”
He added: “I don’t want to even give any impression of trying to impinge on their independence.”
Mr Hancock said the Pfizer/BioNTech application to the US Food and Drug Administration had “given us the confidence formally to start the process here”.
He said by law this first required the Government to ask the MHRA to consider authorisation of a vaccine.
Professor Jonathan Van-Tam, England’s deputy chief medical officer, said the assessment “will happen at the speed of science and we need to leave them to it and trust them to get on with it”.
The NHS is establishing vaccination centres across the UK – ready to roll out a vaccine, if approved, from next month. pic.twitter.com/jIGwqi8ntm
— Matt Hancock (@MattHancock) November 20, 2020
– How will a vaccine be rolled out?
The UK Government has secured 40 million doses of the Pfizer/BioNTech vaccine, with about 10 million of these to arrive before the end of this year.
It has also secured an “initial agreement” for five million doses of the Moderna vaccine if it is approved.
Authorities in the UK have placed orders for 100 million doses of the Oxford vaccine – enough to vaccinate most of the population – should it receive regulatory approval.
Mr Hancock has said the military and NHS staff were on standby to roll out a vaccine across the UK from the start of December, and will work “seven days a week” to do so.
It will be delivered through care homes, GPs and pharmacists, as well as “go-to” vaccination centres set up in venues such as sports halls.
Mr Hancock said the UK will be among the first countries in the world able to do this.
– Are there any logistical issues?
Mr Hancock said Oxford’s vaccine was easier to deploy than Pfizer’s, which needs to be kept at a temperature of minus 70C and is being manufactured in Belgium.
He said that from the moment the Pfizer vaccine leaves the factory in Belgium it can only be taken out of minus 70C four times before it is injected into a patient’s arm.
While it will be a “mammoth logistical operation”, Mr Hancock said he had “confidence” it can be delivered.
– Is the vaccine safe?
All vaccines undergo rigorous testing and have oversight from experienced regulators.
Moderna said its available safety data does not indicate any significant safety concerns.
Their trials found the vaccine was generally safe and well tolerated, and the majority of adverse events were mild or moderate in severity.
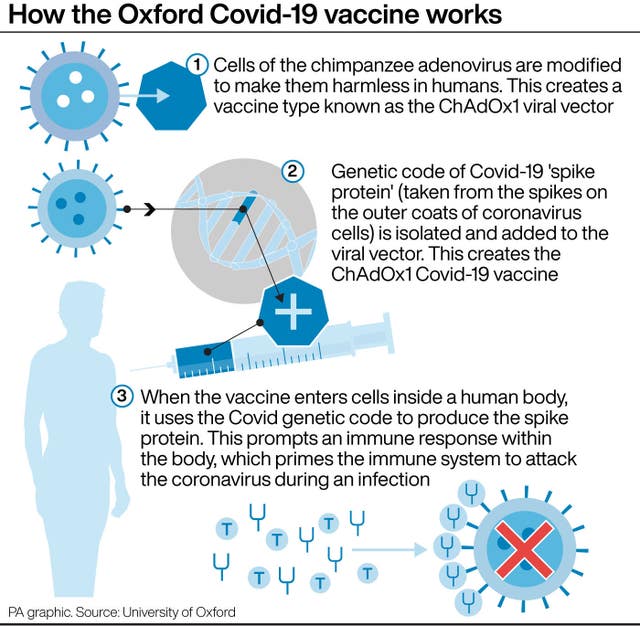
The Pfizer jab is known as a messenger RNA (mRNA) vaccine.
Conventional vaccines are produced using weakened forms of the virus, but mRNAs use only the virus’s genetic code.
They work by telling cells in the body to create antigens, which are recognised by the immune system and prepare it to fight coronavirus.
Some believe mRNA vaccines are safer for the patient as they do not rely on any element of the virus being injected into the body.
The Pfizer vaccine has been tested on 43,500 people in six countries and no safety concerns have been raised.
With the Oxford/AstraZeneca vaccine – known as ChAdOx1 nCov-2019 – no serious adverse health events related to the jab have been seen in the trial participants.
– Who gets a vaccine first?
The Joint Committee on Vaccination and Immunisation (JCVI) has examined data on who suffers the worst outcomes from coronavirus and who is at highest risk of death.
Its interim guidance says that older adults in care homes and care home workers should get priority, followed by those aged 80 and over, and health and social care workers.
Leaked NHS documents, seen by the Health Service Journal (HSJ), apparently suggest all adults in England – of any age – could start to be vaccinated against Covid-19 before the end of January if supplies allow.
Under the plan, every adult who wants a jab could be vaccinated by early April, the HSJ said.
Professor Andrew Pollard from @UniofOxford has been at the forefront of developing a new #COVID19 vaccine.
Watch below to learn about how the Oxford #COVID19 vaccine works ?
Tap the link to share the full video with family and friends on WhatsApp ?
▶️ https://t.co/4EwEo6zP4K pic.twitter.com/4estRGY9O7
— Department of Health and Social Care (@DHSCgovuk) November 20, 2020
– What other vaccines are there?
There are more than 200 coronavirus vaccine candidates being tested around the world.
About 12 of them are in the final stages of testing, but Pfizer is the first to report any results.
Other potential vaccines in phase three trials include one by biotech company Novavax.
– Does this mean life will return to normal soon?
Health Secretary Matt Hancock has said that he has grown “more and more confident” that life will be closer to normal by spring.
Referring to England’s chief medical officer Professor Chris Whitty, who previously said he hopes for a return towards more normality by spring, Mr Hancock said: “And with this (vaccine) news we’ve had over the last few weeks and with the expansion of mass testing I’m more and more confident that he’s right.”
However, Sir John Bell, regius professor of medicine at Oxford University and a member of the Government’s vaccine taskforce, has previously said there was a chance of normality starting to resume after Easter provided “they don’t screw up the distribution of the vaccine”.
Meanwhile, David Nabarro, co-director of Imperial College London’s Institute of Global Health Innovation, said: “Even if a vaccine arrives in the near future we’ve got many months of still dealing with the virus as a constant threat that we’ve got to make certain that we continue to do all that is necessary to solve the virus causing major problems.”